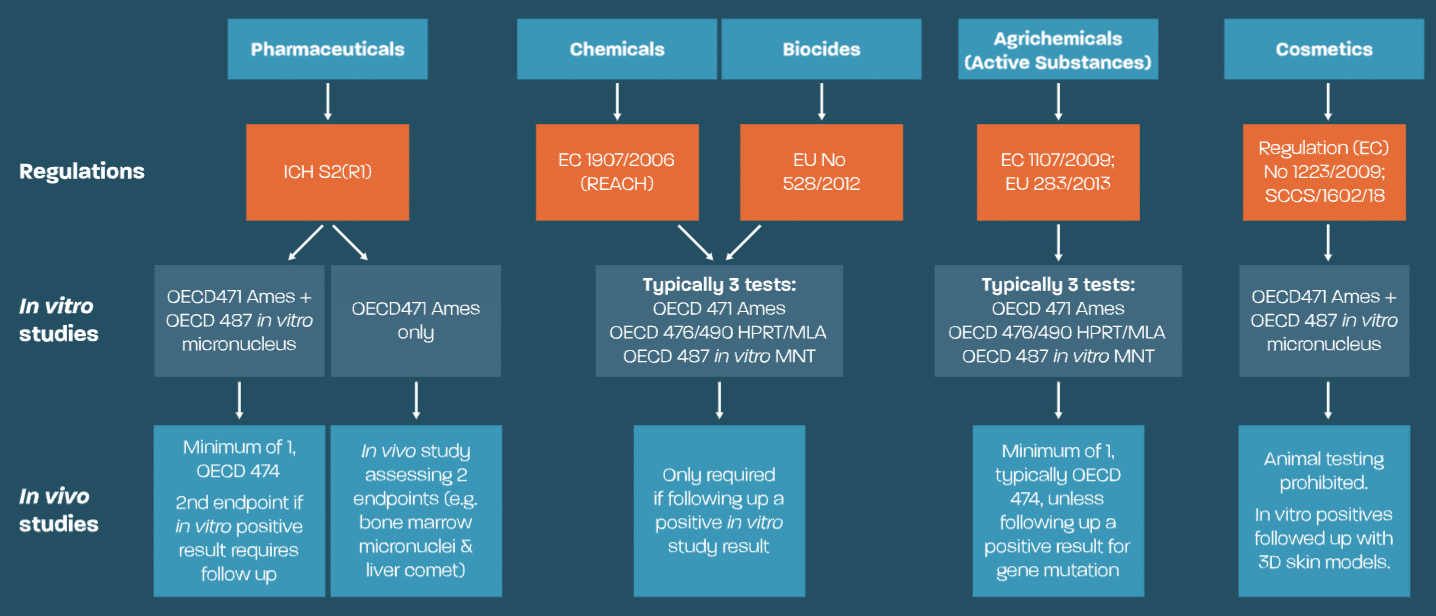
Scantox offers GLP OECD regulatory genotoxicity studies for assessing chemical safety across global industries, covering endpoints detection for mutagenic, clastogenic, and aneugenic mechanisms in our GLP genotoxicity lab.
Service categories:
Comprising a scientific team of experts from pharmaceutical, agrochemical, and CRO backgrounds, Scantox has a wealth of experience in delivering regulatory genotoxicity testing that adheres to genotoxicity studies guidelines. Our laboratory facilities at Alderley Park are GLP-compliant and purpose-designed for genotoxicity testing. We are committed to providing high-quality genotoxicity services and facilitating safety assessment for gene mutation structural and numerical chromosomal damage.
- GLP OECD 471 Ames Test
- OECD 490 GLP Mouse Lymphoma Assay (MLA)
- OECD 476: HPRT Assay
- OECD 474: In Vivo Micronucleus Test
- OECD 488: Big Blue® Transgenic Rodent Somatic and Germ Cell Gene Mutation Assay Test
- Protected: OECD 488: Big Blue® Transgenic Rodent Somatic and Germ Cell Gene Mutation Assay Test
- OECD 489 Comet Assay
- OECD 432: Phototoxicity Test


