Assessing safety, local tolerance and efficacy for wound dressings, pharmaceuticals and combination products
Since 2004, Scantox has conducted wound healing studies for clients developing medical devices, pharmaceuticals and combination products intended for topical application to wounded skin. Studies are performed using Göttingen Minipigs — widely regarded as the most clinically relevant preclinical model for dermal wound healing research.
Why the Göttingen Minipig?
Porcine skin shares several anatomical and functional characteristics with human skin that make it a stronger translational model than rodents. Key similarities include epidermal thickness (70–140 µm in pig versus 70–120 µm in humans), an identical number of epidermal cell layers, sparse hair cover, firm attachment to underlying structures, and the presence of rete ridges. Critically, both pigs and humans heal primarily through re-epithelialization rather than wound contraction — the dominant mechanism in rodents — making the minipig the appropriate species for evaluating treatments that target this phase of healing.
Wound Models Available
Scantox performs four wound model types, each suited to different clinical contexts and product types:
Split-thickness wounds
Split-thickness wounds involve removal of the epidermis and upper dermis using an electrodermatome, creating a superficial lesion that heals by re-epithelialization. Wounds typically close within approximately seven days, making this model well-suited to dressings and formulations targeting superficial skin injury.
Full-thickness wounds
Full-thickness wounds involve removal of epidermis, dermis and subcutis using a circular knife or scalpel. Healing occurs through granulation tissue formation followed by re-epithelialization, with closure typically achieved within approximately three weeks. This model is appropriate for products targeting open wound management. Where relevant, treatment can continue via alternative administration routes including subcutaneous or dermal delivery.
Standard study design involves three animals (approximately 30 kg), with up to eight wounds per animal, allowing up to four treatments to be evaluated simultaneously with each treatment represented across six wounds. This within-animal design improves statistical power while reducing animal numbers.
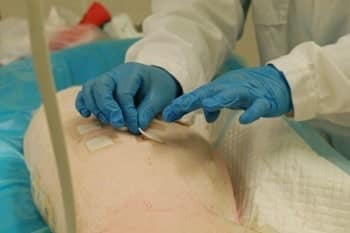
Evaluation Parameters
Wounds are assessed at each dressing change using macroscopic scoring for haemorrhage, wound secretion, inflammation, necrosis, granulation and hypergranulation. Planimetric assessment measures two-dimensional wound area at each timepoint, capturing total wound area, area of granulation tissue, area of re-epithelialized tissue and area of necrotic tissue. Wound outlines are traced on sterile transparent sheets or via digital photography; AI-assisted image analysis using Visiopharm or equivalent software is also available for automated morphometric assessment.
Additional endpoints including tensile strength measurement and laser Doppler skin blood flow assessment can be incorporated based on study objectives and product claims.
Histopathology
Histopathological evaluation is a core endpoint in all Scantox wound healing studies. A single central section through each wound is examined by our senior toxicopathologist, with normal skin on both sides included for comparison. Standard H&E staining of FFPE samples is routinely employed, with special stains (Masson Trichrome, Picrosirius Red) or immunohistochemistry for von Willebrand factor (angiogenesis marker) applied on indication.
Microscopic findings are recorded using a semiquantitative five-grade scoring scheme within the Provantis pathology module, with a detailed narrative report prepared for each study. Our pathologists take a flexible, context-based approach to scoring rather than applying rigid pre-defined numeric systems — consistent with current Society of Toxicologic Pathology guidance, which highlights the limitations of fixed composite scoring protocols (such as ISO 10993-6 Annex E) for wound healing endpoints.
Regulatory Context
No single guideline directly addresses wound healing studies. Relevant frameworks include the EMA Guideline on non-clinical local tolerance testing of medicinal products (EMA/CHMP/SWP/2145/2000 Rev.1) and ICH M3(R2) for safety assessment. Scantox routinely supports study design discussions with sponsors to align study endpoints with regulatory expectations across EMA and FDA pathways.
Studies can be conducted under GLP or non-GLP conditions depending on the stage of development and intended submission purpose.


