In 2006, aggregates and mislocalization of the RNA binding protein TDP-43 were discovered in human amyotrophic lateral sclerosis (ALS) brain samples by Neumann and colleagues. Since then, in vitro and in vivo overexpression of TDP-43 were shown to cause ALS pathology and thus providing a suitable model to study underlying ALS mechanisms. However, transgenic ALS mouse models show a rapidly progressing and fatal pathology. For this reason, we established an inducible mouse model with a moderate progression by injecting an adeno-associated virus of serotype 9 (AAV9) expressing human TDP-43 (hTDP-43) in the motor cortex of adult C57Bl6 mice. Constant expression of hTDP-43 mRNA and protein was confirmed 3-, 6-, and 9-months post injection as assessed with RT-qPCR as well as histological techniques (Figure 1A, B). In more detail, quadruple immunofluorescent labeling revealed that almost exclusively neurons, including CTIP2-positive neurons of the cerebral cortex were transduced, while GFAP-positive astrocytes were TDP-43-negative (Figure 1C).
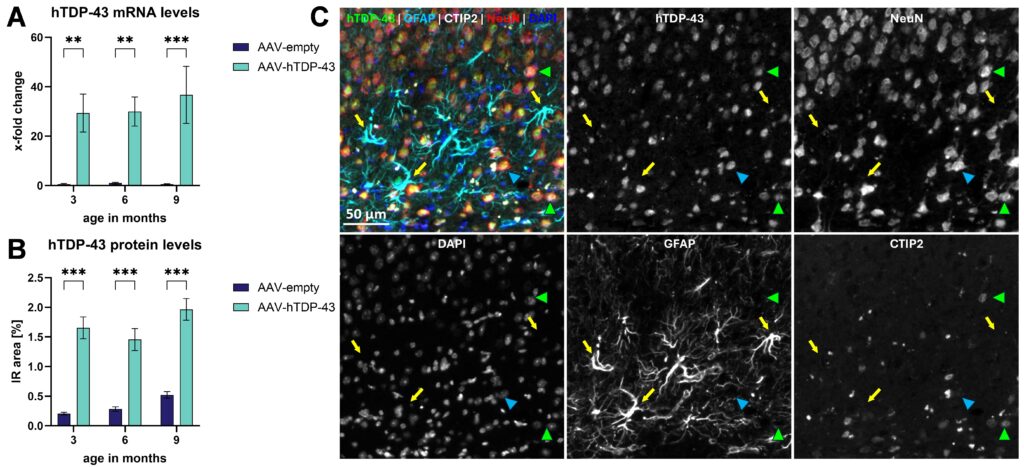
Figure 1: hTDP-43 mRNA and protein expression profile in AAV-hTDP-43-injected mice. Levels of hTDP-43 mRNA (A) and protein (B) in the motor cortex after AAV-hTDP-43 versus AAV-empty control injection. Representative 5-channel image of hTDP-43 (green), GFAP (turquoise), CTIP2 (white), NeuN (red) and DAPI (blue) and single channel images of the same markers (white) in the motor cortex of an AAV-hTDP-43-injected animal. hTDP43 signal co-localizes with NeuN including CTIP2-positive cells (green arrow heads) but not with GFAP-positive astrocytes (yellow arrows), indicating a pure neuronal expression of hTDP-43 (arrow heads). Two-way ANOVA followed by Bonferroni’s post hoc test. Mean ± SEM. **p<0.01; ***p <0.001. n = 8 per group.
AAV-hTDP-43 injection further resulted in a strong increase in GFAP signal 3-, 6- and 9-months post injection using quantitative immunofluorescent image analysis (Figure 2A). A detailed evaluation of the GFAP signal showed an increased number of GFAP-positive cells and these cells were larger and expressed more GFAP per cell compared to cells of the control group (data not shown, contact us for details). These results indicate an increase in the total number of astrocytes and an activation of such astrocytes due to the expression of TDP-43 that stays stable even 9 months after injection.
Additionally, the motor cortex area was significantly smaller in AAV-hTDP-43-injected animals compared to control animals 6- and 9-months post injection; a trend for this effect was already visible 3 months after the application of AAV-hTDP-43 (Figure 2B).
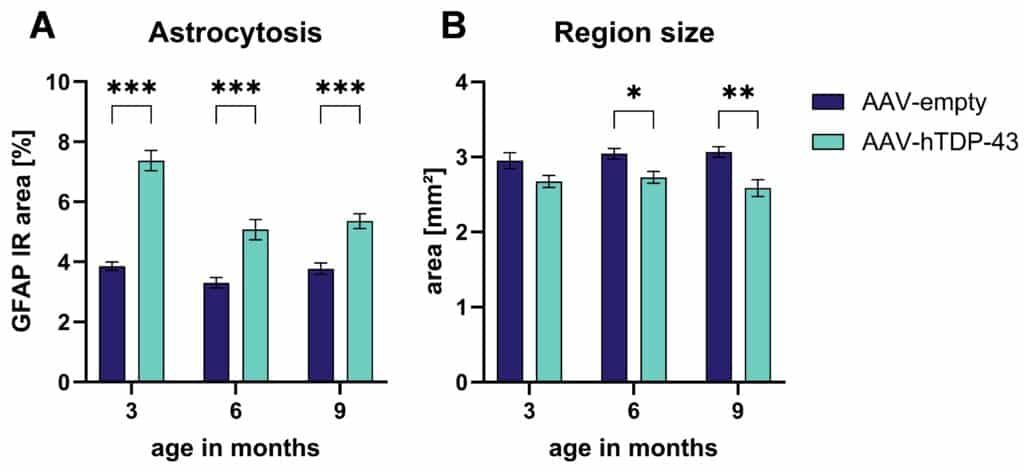
Figure 2: Astrogliosis in and atrophy of the motor cortex in AAV-hTDP-43-injected mice. Quantification of the GFAP immunoreactive (IR) area (A) and the region size (B) of the motor cortex area 3, 6, and 9 months after AAV-hTDP-43 versus AAV-empty control injection. Two-way ANOVA followed by Bonferroni’s post hoc test. Mean ± SEM. *p<0.05; **p <0.01; ***p <0.001. n = 8 per group.
In conclusion, we established a new inducible hTDP-43 model by injecting AAV-hTDP-43 into the motor cortex of adult C57Bl6 mice. The viral injection resulted in the hTDP-43 transduction of only neuronal cells, neuroinflammation as observed by astrogliosis as well as motor cortex atrophy. Consequently, the model can be used for basic research as well as for drug development studies.
Contact us today to get your next ALS study in AAV-hTDP-43-injected mice started.









