Today, over 55 million people worldwide are living with Alzheimer’s disease (AD). While AD is the most common form of dementia, there is currently no cure for the disease. But a recent discovery about the role tau may play in AD represents an exciting breakthrough in our understanding of the progression of AD and other dementias. What is tau, and what does it have to do with neurodegenerative diseases like AD? And how might new discoveries about tau help researchers in the ongoing quest to find a cure for AD? Read on to learn more.
What Is Tau?
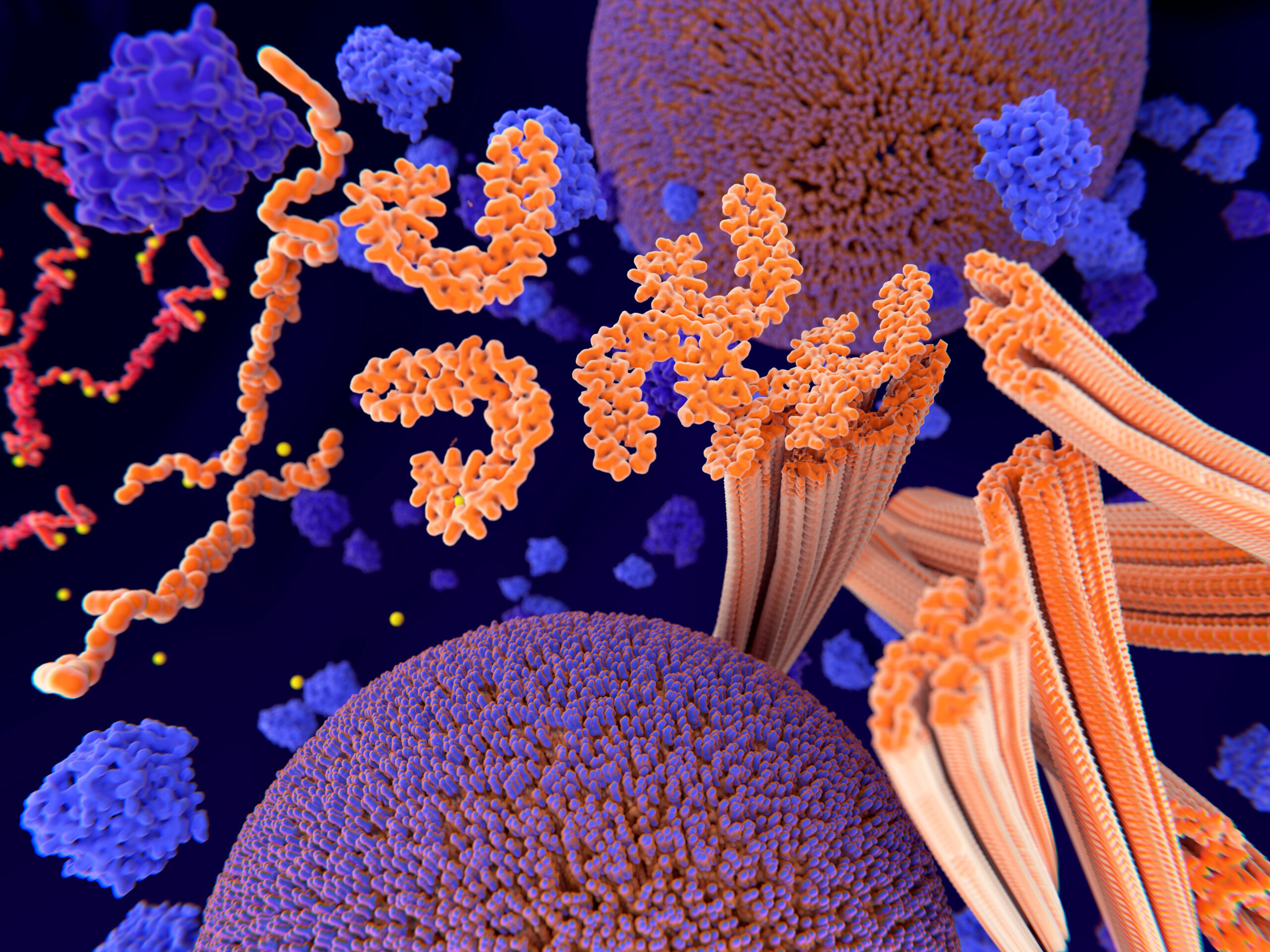
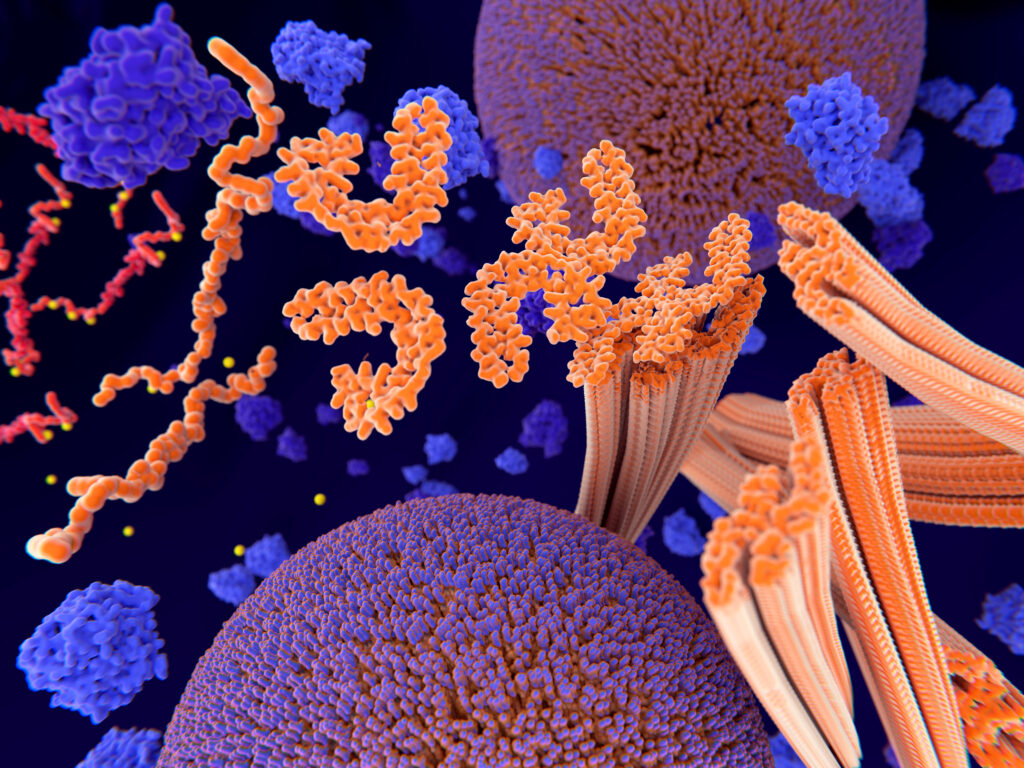
Tau is a protein found mostly in brain cells. In healthy central nervous systems, it works to help stabilize the neurons in the brain, serving structural and regulatory functions that support both axonal transport and signaling.
Malformed Tau and Neurocognition
The link between tau and cognitive function is well-established: studies of mice genetically designed without the tau protein find that cognitive functioning is impaired. And in human studies, tau dysfunction is known to be present in a number of neurodegenerative diseases, including frontotemporal dementia with parkinsonism-17, Pick disease, progressive supranuclear palsy, corticobasal degeneration, chronic traumatic encephalopathy, and AD.
In severe neurodegenerative diseases like the above, malformed tau no longer holds together: instead, it breaks apart, and the disorganized tau strands reform and mutate as malformed, folded, or spliced tau forms that can no longer carry out their physiological function. Weakened, tangled, mutated tau strands instead begin to “wreak havoc” on neurons.
Tau and Alzheimer’s
While it has long been hypothesized by some researchers that plaque build-up of another protein, ß-amyloid, is what causes slowed processing and brain cell death in AD patients, other researchers have suspected that excessive build-up of tau may in fact be the culprit. This is in part because beta amyloid is known to accumulate at the early stages of the disease, while tau accumulation occurs throughout the progression of the disease, from mild to end stages. Scientists have previously found a link between the levels of malformed tau in the brain and the stage and severity of AD. But a new discovery by AD researchers may shed even more light on the role tau plays in AD.
Exploring Tau Hot Spots
New super-resolution microscopy has allowed scientists at the Queensland Brain Institute’s Clem Jones Center for Aging Dementia Research (CJCADR) to observe how individual tau molecules behave at the plasma-membrane barrier. They observed a phenomenon that the team’s co-leads, CJCADR director Professor Jürgen Götz and Dr. Padmanabhan, dubbed “tau hot spots.”
As Dr. Padmanabhan explains, these hot spots are formed as tau localizes at the plasma membrane, where it first displays “highly heterogeneous behavior in space and time.” Once localized, these tau molecules accumulate to form a “hot spot,” or a spot “in which tau molecules are highly immobilized.”
These newly observed tau hot spots could have far-reaching implications for understanding both how AD starts and how it spreads.
Understanding Tau and AD
While the discovery of tau hot spots represents an exciting breakthrough in our ongoing understanding the genesis and progression of AD and other dementias, there is still very little known about the function of tau hot spots in AD. But this new discovery is an important starting point. As Professor Götz explains, “This new research opens the way for gaining deeper insight into how tau and other key proteins impair neuronal functions in a pathological context such as Alzheimer’s disease.”
Scantox is a part of Scantox, a GLP/GCP-compliant contract research organization (CRO) delivering the highest grade of Discovery, Regulatory Toxicology and CMC/Analytical services since 1977. Scantox focuses on preclinical studies related to central nervous system (CNS) diseases, rare diseases, and mental disorders. With highly predictive disease models available on site and unparalleled preclinical experience, Scantox can handle most CNS drug development needs for biopharmaceutical companies of all sizes. For more information about Scantox, visit www.scantox.com.